3. Glass of contaminated water
|
The first sample studied is a liquid collected in a vessel. It is supposed to contain water from the dispenser next to it, but it seems to be extremly contaminated. There are several mixed substances. Some of them are seen with the naked eye such as pencil shavings, metal clips, sand and oil; But we do not know if there is anything more dissolved in the water. The only element that could be the cause of the heart attack of John Carles is the salt; will there be salt in the glass? How will we isolate it? Let's go to the lab!
|
ACTIVITY 5:
Working in groups, we analyze the mixture in the laboratory and separate its components. We will fill out this SHEET of the separation methods used and the results, completing it with photographs or videos of the process. The report will be evaluated with a corresponding rubric.
Working in groups, we analyze the mixture in the laboratory and separate its components. We will fill out this SHEET of the separation methods used and the results, completing it with photographs or videos of the process. The report will be evaluated with a corresponding rubric.
|
LABORATORY
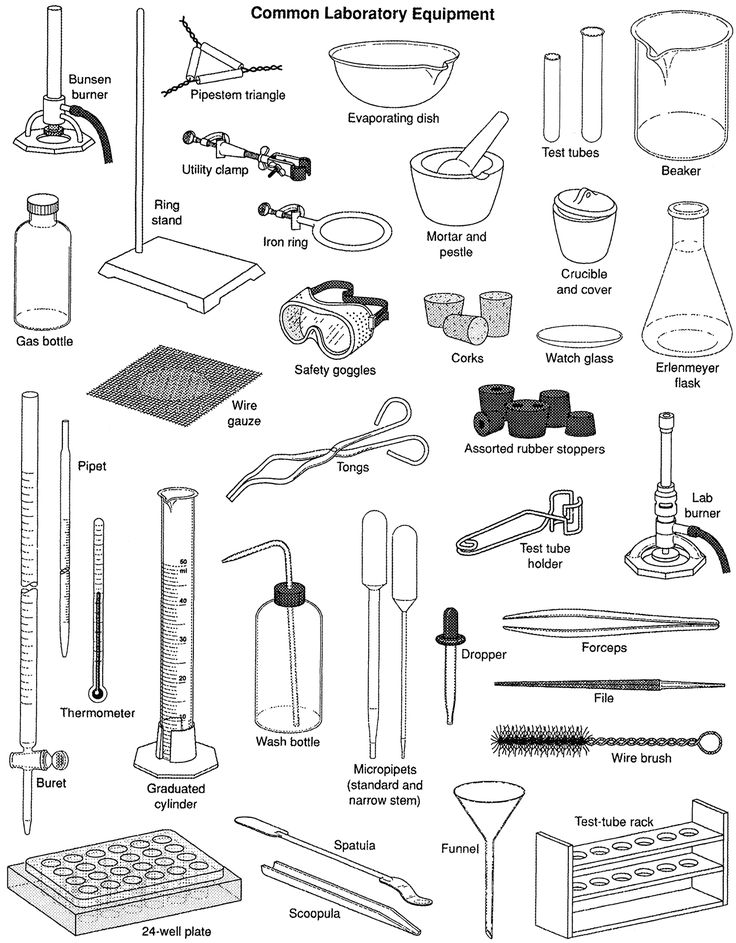
Materials: Mixing problem (250 ml of water, substance dissolved in saturation, pencil shavings, sand, 25 ml of oil, metal clips) Beakers Flasks Crystalizer Petri plates or watch glasses Funnels Glass stirrer or spatulas Decantion funnel Pipettes Laboratory stands Pliers Filter paper Magnet Strainer or sieve Process: 1) Separate the metal clips with a magnet. Attach a magnet to the mixture that will attract the clips. |
2) We make a separation of the pencil chips by sieving. We simply pick them up with a small kitchen strainer (or proper pore sieve).
3) Next, we separate the sand by filtration. Let the mixture rest and pour into another glass, very carefully, the part where the oil is (so that it does not hinder the filtration). Then we take a flask and put a funnel and a filter paper in his mouth. Then we stir the mixture of the glass with a stirrer and filter it through the paper filter. In it will remain the sand and in the flask the rest of our sample, which we incorporate the part with oil, to continue working with it.
4) Now, we will have a visible mixture of water and oil. Separate the oil by decantation. We make the assembly with the decanting funnel and pass the mixture to the funnel. We open the key little by little until we separate the water that we will collect in a beaker.
5) Now we have a homogeneous sample, where, in principle, we do not appreciate if there is something dissolved in the water. Will there be salt? To know, we take a few ml. With a pipette and pour them into a Petri dish or a crystallizer (separation by crystallization). We will have to wait a few weeks to see if, when the water evaporates, there is some residue of salt that was dissolved.
Perfect! We prepared for the next sample, the suicide note. Let's go to the beginning and press number 4
Complementary activities
If you want to get extra points, individually or by forming a group with other colleagues, you can do these activities
- Make a column of densities
- Crystallizations